The Orange Cap Problem
A record endotoxin result, repeated failures, and a batch label mess that turned JEEP T30 into a market-wide warning
After the SS-31 warning, a new alert began circulating through the grey-market peptide community and across multiple forums on April 8. This time, the focus was JEEP’s orange-cap T30.
An orange-cap T30 batch purchased in January 2026 first returned an estimated endotoxin result of 1350 EU/vial. To check the accuracy of that result, two further vials from the same batch, but donated by different users in different countries were sent for testing. One returned a second estimate of 1360 EU/vial. Another, tested at a higher dilution, returned a result of 2306 EU/vial, the highest reported result ever so far according to Janoshik.
That left three vials from the same batch testing at unacceptably high endotoxin levels.
Not just one result
The issue was complicated further by the appearance of two different shades of orange cap.
Reports suggested the lighter orange caps are the affected ones, while the darker orange caps, in circulation prior appeared unaffected. At the same time, a newer black-cap batch of T30 from Jeep has not yet been fully tested, but is already being sold for $98 per kit.
There were also anecdotal reports of rashes and increased heart rate. The advice being shared was simple: anyone who purchased this batch should not use it.
A separate sample later sent to TrustPointe reportedly returned 110 EU, adding another layer to the endotoxin picture rather than resolving it.
Three vials. Repeated high results. One record figure. By that point, this was no longer a one-off anomaly.
Sterility is not the same as endotoxin
I recently wrote in detail about endotoxin contamination, so I won’t repeat everything here. But the distinction matters.
Sterility testing looks for live bacteria.
Endotoxin testing looks for bacterial fragments, specifically lipopolysaccharides (LPS).
These endotoxins are toxic components found in the outer membrane of gram-negative bacteria such as E. coli. Crucially, they can remain in a product even after the bacteria themselves have been killed.
That creates two separate issues.
First, the clinical impact. Endotoxins can cause fever, chills, rashes, and increased heart rate as well as the potential to trigger immune reactions. These effects can worsen with repeated exposure, as endotoxin load builds over time.
Second, what their presence signals. Endotoxins are not random. They point to failures somewhere in the manufacturing process.
Typical sources include poor water quality control, inadequate cleaning procedures, contamination during handling or vial filling, and weak overall process control.
Endotoxin contamination is not just a safety issue. It is a quality-control failure.
What do the official limits actually say?
This is where a lot of forum discussion goes sideways, because people throw around raw EU numbers without grounding them in the actual compendial framework.
For most non-intrathecal injectable drugs, the standard reference value used in endotoxin limit calculations is 5 EU/kg body weight per hour. That does not translate into one simple universal “safe number” per vial, because endotoxin limits are tied to dose. But it does show why results of 1350–1360 EU/vial, and especially 2306 EU/vial, were treated as a serious warning rather than a minor technical quibble.
From vendor issue to market-wide concern
Once it became clear that orange-cap T30 was affected, attention quickly shifted beyond JEEP.
People began checking other T30 they had purchased. If JEEP sourced from the same manufacturer as other vendors, the question was no longer just whether one batch had failed, but whether the issue could extend further upstream.
A recent COA from MIX Peptides indicated they had also sold orange-cap T30 around the same time. Other vendors known to have sold orange-cap T30 include Uther, Allen ABC, and KBR (Kepai Beauty Research).
Uther is a lyophiliser himself. If his are also found to be affected, that would point more strongly to a problem at the raw supply level rather than only at the finishing stage.
Samples from Uther and Allen ABC have now been sent for testing.
At this stage, there are no confirmed endotoxin results yet from those vendors. But because many sellers source from the same small group of factories, the possibility of wider overlap is now being taken seriously.
What initially looked like a batch issue may in fact be a supply-chain issue.
If that turns out to be the case, then the real problem is not just one vendor’s bad lot. It is the possibility that the same contaminated material was distributed across multiple sellers.
Then came the batch-label mess
As if the endotoxin results were not bad enough, the traceability picture quickly became murkier.
Questions emerged over:
reused batch numbers
mismatched cap colours
manufacture-date contradictions
customers being shown reports that did not clearly match the product in hand
That matters because once a contamination warning is live, buyers need to know one thing above all else:
Do I actually have the affected product or not?
Instead, what emerged was confusion.
Some buyers were told their product was from a different batch, only to be shown reports tied to labels or cap colours that did not line up neatly with manufacture dates. Others pointed to older and newer stock carrying the same batch numbering pattern. The result was predictable: confidence in the label trail started to collapse.
A contamination problem is bad. A contamination problem plus broken traceability is worse.
What is JEEP going to do?
The next question on everyone’s lips was what JEEP was actually going to do about it.
Responses from Chinese vendors are not always immediate, partly because of the time difference. But there was another issue too: most grey-market vendors explicitly guarantee only mass and purity. They do not generally guarantee sterility or endotoxin levels.
That leaves vendors in a familiar grey zone: commercially, they may claim they owe buyers nothing. Morally, that is another matter.
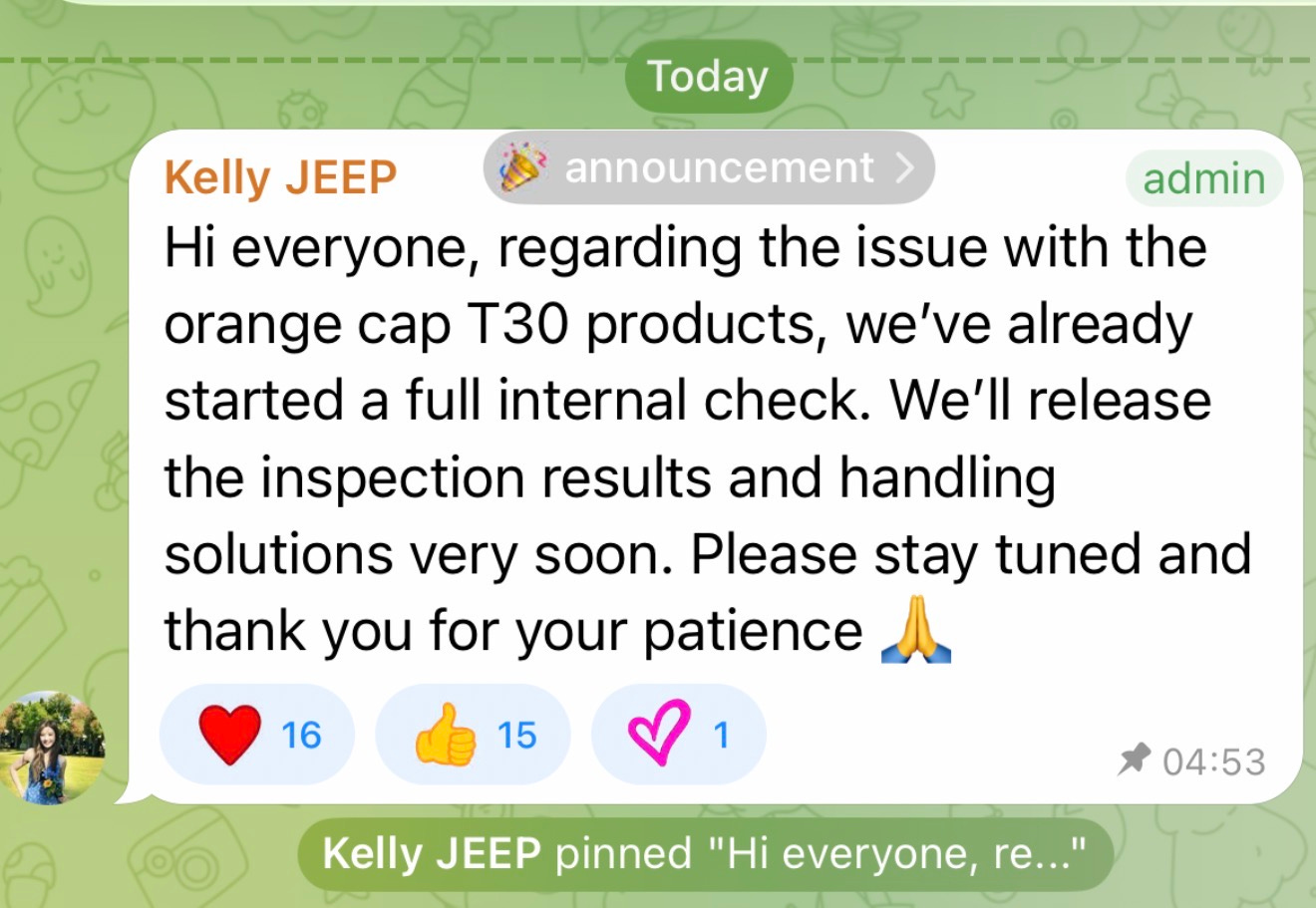
On 8 April at 04:53 UK time, Kelly JEEP addressed the issue publicly. She said they had started a full internal check and would release the inspection results and proposed handling solutions soon.
For now, the investigation is ongoing.
Where this leaves the market
This is no longer just a question of one vendor or one batch.
It is a question of:
traceability
manufacturing controls
supplier overlap
and how much trust can realistically be placed in a fragmented grey-market supply chain
What started as a single high endotoxin result has now escalated into something larger:
A record-level contamination result, repeated across samples, with no clear boundary yet on where it begins or ends.
And that is why orange-cap T30 stopped being just another product line and became a market-wide warning.
To be continued …….







I appreciate this thread very much. I don't use t30 but if I did I'd be concerned too
First of all Krysia's explanation of how it could have happened to cause the endotoxin problem was so good that I have nothing to add. This was obviously a lab mishap.
Barn orange cap T-30 sold at about the same time passed endotoxin testing. This tells me that not all orange cap T-30 are contaminated. They do look dark orange with silver crimps. GYC is the same color cap and crimps but were not tested for endotoxins.
I have been saying for years that cap colors and batch numbers are almost meaningless. All the dealers are buying from the same handful of factories. There is no way of knowing for sure if they are all from the same batch. A factory will make 10-20 thousand vials at a time before cleaning everything up for the next batch. There could be 100s of thousands of orange cap T-30 vials out there. Batch numbers don't mean anything since dealers will often use the same numbers on every kit that comes in as long as cap colors match up.